In vivo and in vitro imaging
Coordinators
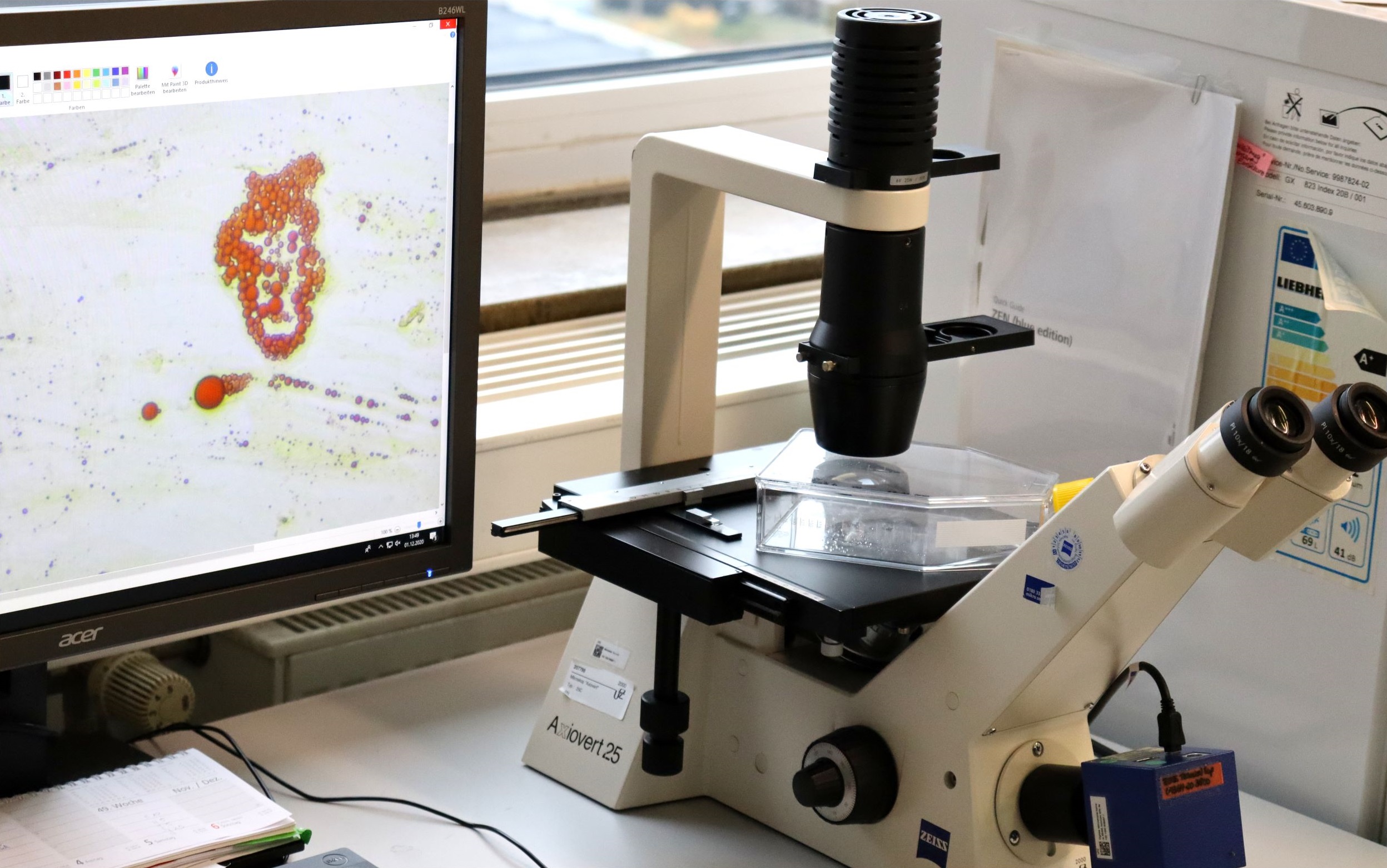
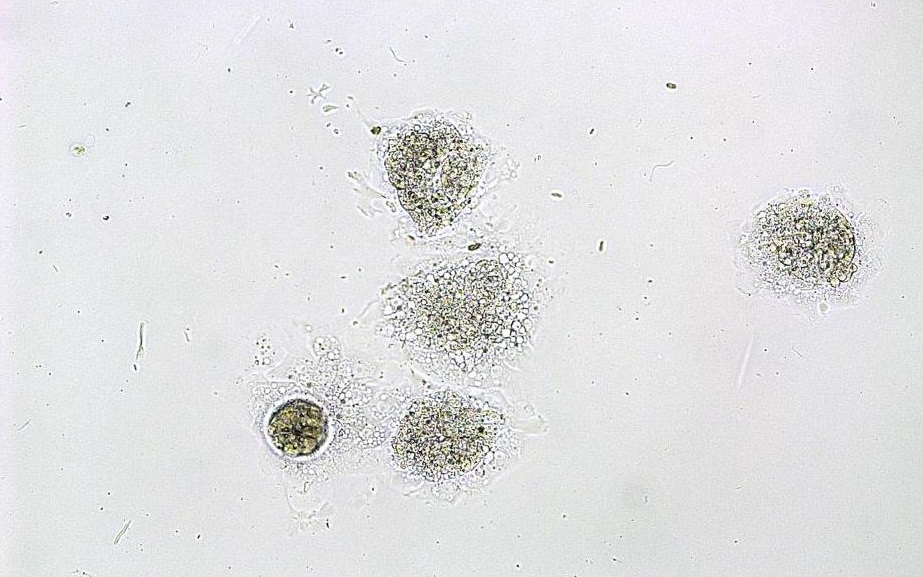
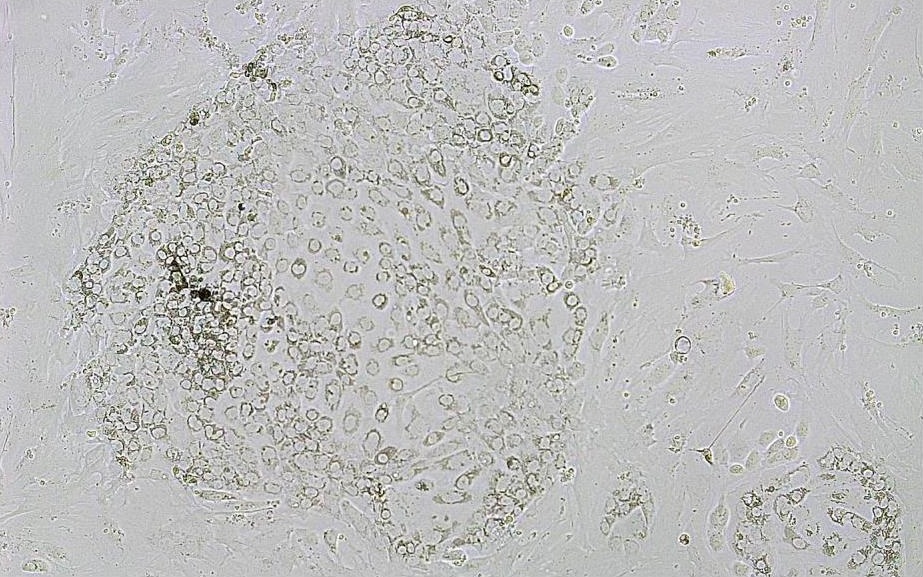
In vitro imaging comprises microscopic images and their processing with the compact fluorescence microscop BZ-X800 (Keyence Corporation, Japan). Besides the simple image display extended analyses, such like
(i) counting and quantification of target areas within an image,
(ii) 3D-analysis,
(iii) time-lapse recordings even under culture conditions (CO2-gassing)
are optional applications.
In vivo imaging can be used for the visualization of biological compounds within living organisms. This is useful to investigate treatment effects, disease or tumor progression, and pregnancy development. During molecular imaging, fluorescent biomarkers are utilized for the visualization of cellular functions and molecular processes. In a physiological relevant manner, time-dependent changes can be analyzed.
In contrast there are anatomical imaging methods, like tomography or sonography/ultrasound. The VEVO 2100 Imaging System (FujiFilm VisualSonics Inc.) is a high frequency ultrasound device (Figure 1), generated for the analysis of small animals.
 Figure 1: The Vevo 2100 Imaging System. Main control unit (A) with heating platform control pad (B), gel warmer (C), isoflurane control unit (D), knock down box (E), heated platform with four copper areas (F; F.1), transducer (G), transducer holding device (H). Figure republished from [3].
Figure 1: The Vevo 2100 Imaging System. Main control unit (A) with heating platform control pad (B), gel warmer (C), isoflurane control unit (D), knock down box (E), heated platform with four copper areas (F; F.1), transducer (G), transducer holding device (H). Figure republished from [3].
It can be used for various research fields like cardiology, development, molecular imaging, oncology, neurobiology, urogenital and vascular biology as well as pregnancy research[1]. Furthermore, the influence of environmental chemicals can be impressively investigated in vivo. Ultrasound is a standard method during pregnancy. Fetal developmental disabilities or impairments can be recognized early. The VEVO 2100 Imaging System enables us to analyze fetuses and placentas in pregnant mice in vivo (Figure 2, 3). Moreover, we can follow–up fetal as well as maternal blood flow parameters throughout pregnancy (Figure 4). The technique is important to assess intrauterine growth restriction, a pregnancy complication with short- and long-term health consequences for both mother and fetus. The high utility of this method is exemplified by its non-invasiveness and the possibility to analyze the same animal throughout pregnancy, without sacrificing several animals at different gestational days[2,3].
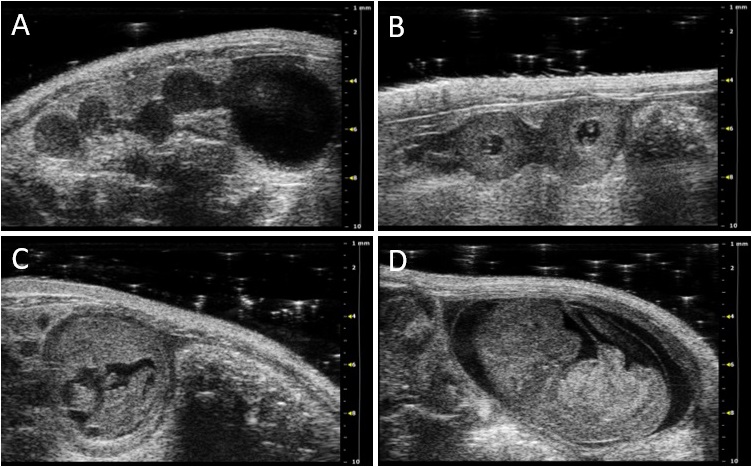 Figure 2: 2D grey-scale ultrasound images showing implantation areas from mice at gestation day (gd) 5 (A), gd8 (B), gd10 (C), and gd12 (D). Figure republished from [3].
Figure 2: 2D grey-scale ultrasound images showing implantation areas from mice at gestation day (gd) 5 (A), gd8 (B), gd10 (C), and gd12 (D). Figure republished from [3].
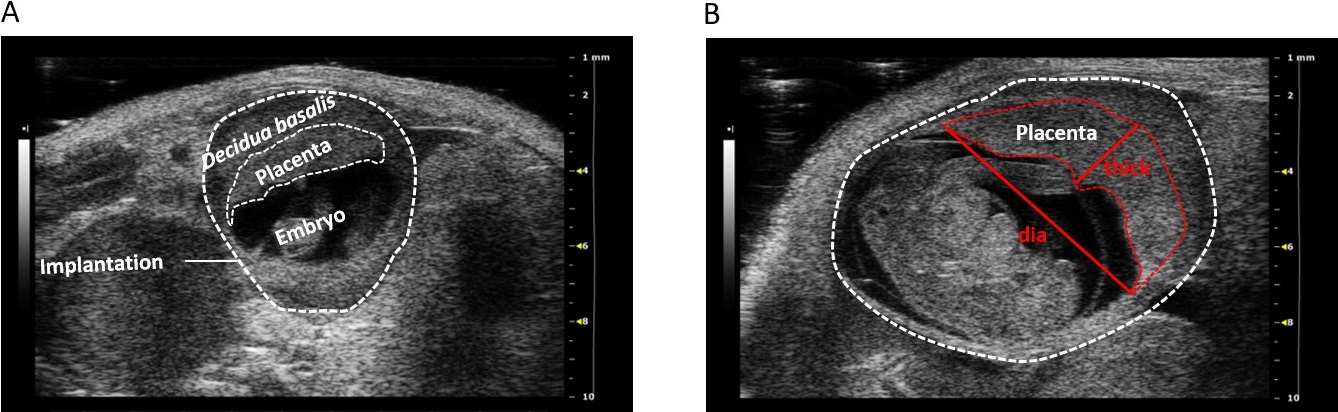 Figure 3: (A) 2D grey-scale ultrasound image of an implantation at gestation day (gd) 10 showing decidua basalis, placenta, embryo. (B) 2D grey-scale ultrasound image of an implantation at gd12 showing placental thickness (thick) and placental diameter (dia). Figure republished from [3].
Figure 3: (A) 2D grey-scale ultrasound image of an implantation at gestation day (gd) 10 showing decidua basalis, placenta, embryo. (B) 2D grey-scale ultrasound image of an implantation at gd12 showing placental thickness (thick) and placental diameter (dia). Figure republished from [3].
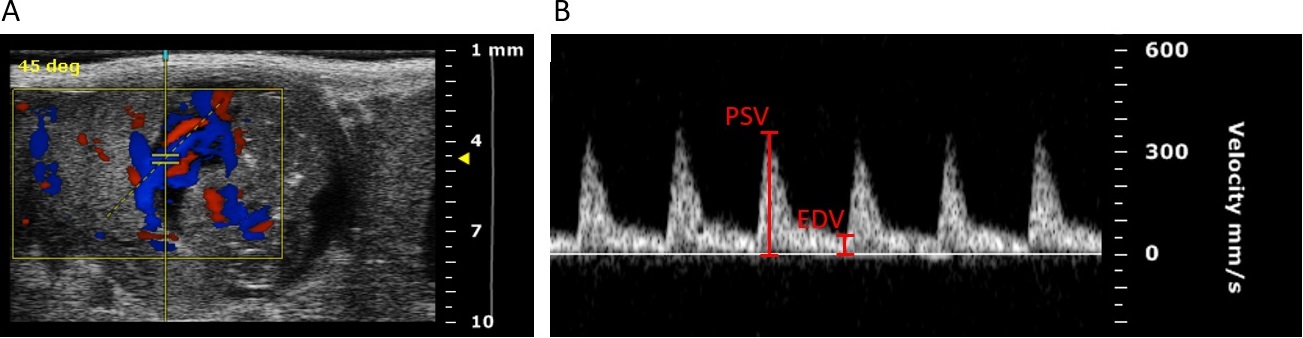 Figure 4: Color Doppler image of a fetal umbilical artery at gestation day (gd) 14. (B) Pulse-wave Doppler images of a maternal uterine artery at gd10 showing peak systolic velocity (PSV) and end diastolic velocity (EDV). Figure is republished from [3].
Figure 4: Color Doppler image of a fetal umbilical artery at gestation day (gd) 14. (B) Pulse-wave Doppler images of a maternal uterine artery at gd10 showing peak systolic velocity (PSV) and end diastolic velocity (EDV). Figure is republished from [3].
References:
[1] FujiFilm VisualSonics Inc. website: https://www.visualsonics.com
[2] Meyer N, Schüler T, Zenclussen AC. Simultaneous Ablation of Uterine Natural Killer Cells and Uterine Mast Cells in Mice Leads to Poor Vascularization and Abnormal Doppler Measurements That Compromise Fetal Well-being. Front Immunol. 2018;8:1913. doi: 10.3389/fimmu.2017.01913. eCollection 2017.
[3] Meyer N, Schüler T, Zenclussen AC. High Frequency Ultrasound for the Analysis of Fetal and Placental Development in vivo. J Vis Exp. 2018;(141). doi: 10.3791/58616.