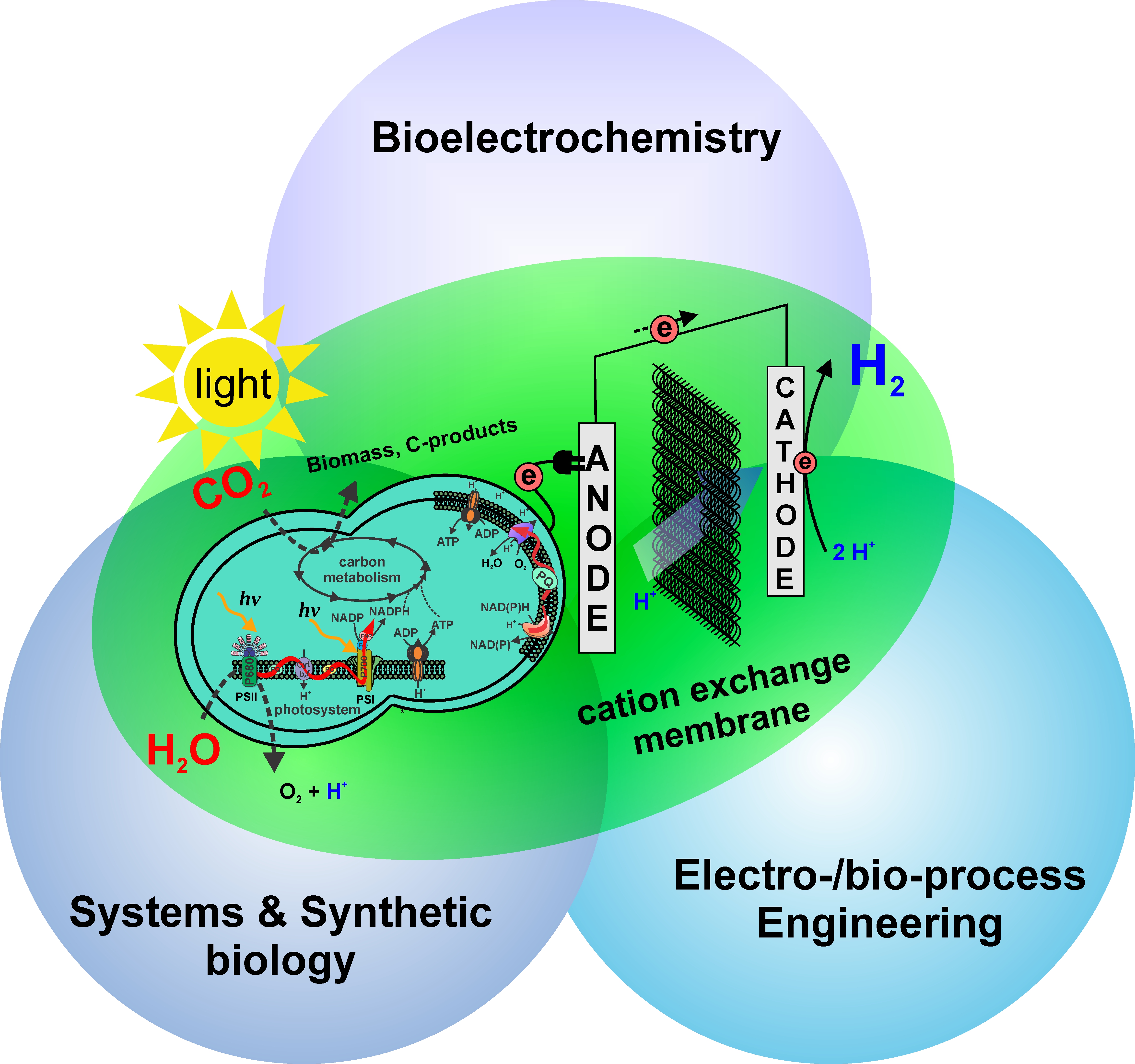
Welcome to the BMBF-funded junior research group Biophotovoltaics.
The research group is funded via the BMBF junior research group funding through the thematic call "Creative young researchers for the bioeconomy". The research group focuses on the biohybrid technique called Biophotovoltaics (BPV) for sustainable energy production from sunlight and water, using interdisciplinary research approach integrating synthetic biology, systems biology, bio- and electrochemical-process engineering and modelling. We are interested in the fundamental, quantitative and holistic understandings of extracellular electron transfer pathway on molecular levels and its corresponding biochemistry and microbial physiology, and subsequently the rational design and development of novel process concepts for optimized desired purpose.
We are always welcome talent, self-motivated and passionate young scientist to join our group. Master and bachelor thesis projects are always available. If you are interested in our work, please contact bin.lai@ufz.de.
Head
Postdoc:
Project: membrane redox protein for extracellular electron transfer of Synechocystis in Biophotovoltaics (BMBF, from 03.2023)
Project: determination of the mass and electron transfer kinetics in a cyanobacterial biofilm (EU JTF, BISON project, from 11.2024)
Project: system optimization of biophotovoltaics system (EU JTF, BISON project, from 11.2024)
PhDs:
Thesis: Electrifying Lactococcus lactis for more sustainable food fermentations (CSC, from 11.2024)
Thesis: Engineering the membrane transportation system of Synechocystis sp 6803 in Biophotovoltaics (BMBF, from 11.2023)
Thesis: Process modelling and computational-guided system development of Biophotovoltaics (BMBF, from 10.2023)
Thesis: process engineering and system development of Biophotovoltaics (BMBF, from 09.2023)
Thesis: electro-microbiology of Synechocystis in Biophotovoltaics (BMBF, from 03.2023)
Research Scientist
Hannah Schön (11.2025 - )
Masters
HIWI
Antonia Breier
Alumnae:
10.2024 - 02.2026: Ms. Ragini Gabhrani (Technician)
05.2025 - 11.2025: Mr. Souvick Maji (Master thesis, Uni Leipzig)
05.2025 - 11.2025: Ms. Maria Veit (Master thesis, Uni Leipzig)
09.2021 - 08.2025: Mr. Jianqi Yuan (PhD, jointly supervised with Prof. Jens Krömer)
04.2024 - 03.2025: Ms. Hannah Schön (Master thesis, TU Dresden)
12.2020 - 10.2024: Mr. Hans Schneider (PhD, jointly supervised with Prof. Jens Krömer)
06.2021 - 10.2024: Ms. Laura Pause (PhD, jointly supervised with Prof. Jens Krömer)
06.2022 - 07.2024: Ms. Maria Veit (Uni Leipzig, Bachelor thesis + HIWIs)
02.2023 - 04.2023: Ms. Liuyan Gu (PhD student at DTU, Denmark)
09.2026 - 02.2029
Funding Agency: Novo Nordisk Foundation
Grant number: NNF25OC0104942 (Photobioammonium: a novel photo-bioelectrochemical hybrid platform for ammonia production)
Budget: c.a. DKK2.15M kr. (out of DKK 6.5M kr. for three partners)
Co-PI: Dr. Bin Lai.
2024.09 - 2026.06
Funding Agency: EU-Förderung Just Transition Fund (JTF) in Sachsen 2021 - 2027
Grant Number: 100702610 (BISON: Biohybrides Sonnenkraftwerk zur Umwandlung von Licht in Chemische Energie – nachhaltig, effizient, ressourcenschonend)
Budget: c.a. 3M €
co-PI: Dr. Bin Lai (WP5 leader)
2023.07
Funding Agency: Chinesisch-Deutsches Zentrum für Wissenschaftsförderung;
Grant number: GT 1725;
Budget: 2245 €
PI: Dr. Bin Lai
2022.08 - 2027.07
Funding Agency: Federal Ministry of Education and Research (BMBF);
Grant number: 031B1273 (BioPV4H2: biophotovoltaics for biohydrogen production from water, carbon dioxide and sunlight)
Budget: 2.910.350,31 €
PI: Dr. Bin Lai
Index:
You could use our publication index for further requests.
2026 (2)
- Li, R., Gu, L., Blank, L.M., Solem, C., Lai, B. (2026):
Electrifying food fermentations to promote sustainability
Trends Biotechnol. 44 (2), 303 - 314
10.1016/j.tibtech.2025.07.001 - Yuan, J., Lai, B., Krömer, J.O. (2026):
Comparative effects of monochromatic red and broad-spectrum white light on biophotovoltaics: Stability, efficiency, and application potential
Bioelectrochemistry 168 , art. 109164
10.1016/j.bioelechem.2025.109164
2025 (6)
- Ji, Q., Liu, Y., Zhang, H., Gao, Y., Ding, Y., Xie, J., Zhang, J., Jin, X., Lai, B., Chen, C., Wang, J., Gao, W., Mei, K. (2025):
Structural insights into the substrate recognition of ginsenoside glycosyltransferase Pq3-O-UGT2
Adv. Sci. 12 (11), art. 2413185
10.1002/advs.202413185 - Schneider, H., Lai, B., Krömer, J.O. (2025):
Understanding the electron pathway fluidity of Synechocystis in biophotovoltaics
Plant J. 121 (2), e17225
10.1111/tpj.17225 - Veit, M.C., Stauder, R., Bai, Y., Gabhrani, R., Schmidt, M., Klähn, S., Lai, B. (2025):
The necessity of multi-parameter normalization in cyanobacterial research: A case study of the PsbU in Synechocystis sp. PCC 6803 using CRISPRi
J. Biol. Chem. 301 (11), art. 110763
10.1016/j.jbc.2025.110763 - Weimer, A., Krömer, J., Lai, B., Wittmann, C. (2025):
The TonB-dependent transport system facilitates the uptake of inorganic metal mediators in Pseudomonas putida KT2440 in a bioelectrochemical system
Microb. Biotechnol. 18 (8), e70206
10.1111/1751-7915.70206 - Yuan, J., Appel, J., Gutekunst, K., Lai, B., Krömer, J.O. (2025):
Molecular dynamics of photosynthetic electron flow in a biophotovoltaic system
Environ. Sci. Ecotechnol. 23 , art. 100519
10.1016/j.ese.2024.100519 - Yuan, J., Bai, Y., Lenz, C., Reilly-Schott, V., Schneider, H., Lai, B., Krömer, J.O. (2025):
The impact of redox mediators on the electrogenic and physiological properties of Synechocystis sp. PCC 6803 in a biophotovoltaic system
ChemSusChem 18 (13), e202402543
10.1002/cssc.202402543
2024 (4)
- Lai, B. (2024):
Burning questions: Exploring the limits of microbial electrochemical technology for industrial biotechnological applications
Microb. Biotechnol. 17 (1), e14370
10.1111/1751-7915.14370 - Pause, L., Weimer, A., Wirth, N.T., Nguyen, A.V., Lenz, C., Kohlstedt, M., Wittmann, C., Nikel, P.I., Lai, B., Krömer, J.O. (2024):
Anaerobic glucose uptake in Pseudomonas putida KT2440 in a bioelectrochemical system
Microb. Biotechnol. 17 (1), e14375
10.1111/1751-7915.14375 - Reilly-Schott, V., Gaibler, J., Bai, Y., Mier-Jimenez, A., Qasim, M., Lai, B. (2024):
Electron leaks in biophotovoltaics: A multi-disciplinary perspective
ChemCatChem 16 (18), e202400639
10.1002/cctc.202400639 - Weimer, A., Pause, L., Ries, F., Kohlstedt, M., Adrian, L., Krömer, J., Lai, B., Wittmann, C. (2024):
Systems biology of electrogenic Pseudomonas putida - multi-omics insights and metabolic engineering for enhanced 2-ketogluconate production
Microb. Cell. Fact. 23 , art. 246
10.1186/s12934-024-02509-8
2023 (6)
- Gu, L., Xiao, X., Lee, S.Y., Lai, B., Solem, C. (2023):
Superior anodic electro-fermentation by enhancing capacity for extracellular electron transfer
Bioresour. Technol. 389 , art. 129813
10.1016/j.biortech.2023.129813 - Lai, B. (2023):
Biophotovoltaik: mikrobielle Wasserstoffproduktion
Biospektrum 29 (1), 97 - 98
10.1007/s12268-023-1873-3 - Lai, B., Krömer, J., Aulenta, F., Wu, H., Nikel, P.I. (2023):
Exploiting synergies between microbial electrochemical technologies and synthetic biology
Microb. Biotechnol. 16 (3), 485 - 488
10.1111/1751-7915.14208 - Schneider, H., Lai, B., Krömer, J. (2023):
Interference of electron transfer chain inhibitors in bioelectrochemical systems
Electrochem. Commun. 152 , art. 107527
10.1016/j.elecom.2023.107527 - Schneider, H., Lai, B., Krömer, J. (2023):
Utilizing cyanobacteria in biophotovoltaics: An emerging field in bioelectrochemistry
In: Bühler, K., Lindberg, P. (eds.)
Cyanobacteria in biotechnology. Applications and quantitative perspectives
Adv. Biochem. Eng. Biotechnol. 183
Springer Nature, p. 281 - 302
10.1007/10_2022_212 - Wang, M., Ji, Q., Lai, B., Liu, Y., Mei, K. (2023):
Structure-function and engineering of plant UDP-glycosyltransferase
Comp. Struct. Biotechnol. J. 21 , 5358 - 5371
10.1016/j.csbj.2023.10.046
2022 (2)
- Gemünde, A., Lai, B., Pause, L., Krömer, J., Holtmann, D. (2022):
Redox mediators in microbial electrochemical systems
ChemElectroChem 9 (13), e202200216
10.1002/celc.202200216 - Lai, B., Glaven, S., Song, H. (2022):
Editorial: Electrobiotechnology towards sustainable bioeconomy: Fundamental, optimization and applications
Front. Bioeng. Biotechnol. 10 , art. 901072
10.3389/fbioe.2022.901072
2021 (3)
- Lai, B., Schneider, H., Tschörtner, J., Schmid, A., Krömer, J.O. (2021):
Inside front cover image, Volume 118, Number 7, July 2021
Biotechnol. Bioeng. 118 (7), ii - ii
10.1002/bit.27411 - Lai, B., Schneider, H., Tschörtner, J., Schmid, A., Krömer, J.O. (2021):
Technical-scale biophotovoltaics for long-term photo-current generation from Synechocystis sp. PCC6803
Biotechnol. Bioeng. 118 (7), 2637 - 2648
10.1002/bit.27784 - Nguyen, A.V., Lai, B., Adrian, L., Krömer, J.O. (2021):
The anoxic electrode-driven fructose catabolism of Pseudomonas putida KT2440
Microb. Biotechnol. 14 (4), 1784 - 1796
10.1111/1751-7915.13862
2020 (3)
- Kadier, A., Jain, P., Lai, B., Kalil, M.S., Kondaveeti, S., Alabbosh, K.F.S., Abu-Reesh, I.M., Mohanakrishna, G. (2020):
Biorefinery perspectives of microbial electrolysis cells (MECs) for hydrogen and valuable chemicals production through wastewater treatment
Biofuel Res. J. 25 , 1128 - 1142
10.18331/BRJ2020.7.1.5 - Lai, B., Bernhardt, P.V., Krömer, J.O. (2020):
Cytochrome c reductase is a key enzyme involved in the extracellular electron transfer pathway towards transition metal complexes in Pseudomonas putida
ChemSusChem 13 (19), 5308 - 5317
10.1002/cssc.202001645 - Lai, B., Krömer, J.O. (2020):
Steering redox metabolism in Pseudomonas putida with microbial electrochemical technologies
In: Tiquia-Arashiro, S.M., Pant, D. (eds.)
Microbial Electrochemical Technologies
CRC Press, Boca Raton, FL, p. 59 - 75
10.1201/9780429487118
2019 (3)
- Cui, Y., Lai, B., Tang, X. (2019):
Microbial fuel cell-based biosensors
Biosensors 9 (3), art. 92
10.3390/bios9030092 - Lai, B., Nguyen, A.V., Krömer, J.O. (2019):
Characterizing the anoxic phenotype of Pseudomonas putida using a bioelectrochemical system
Methods Protoc. 2 (2), art. 26
10.3390/mps2020026 - Tschörtner, J., Lai, B., Krömer, J.O. (2019):
Biophotovoltaics: green power generation from sunlight and water
Front. Microbiol. 10 , art. 866
10.3389/fmicb.2019.00866
2018 (2)
- Kracke, F., Lai, B., Yu, S., Krömer, J.O. (2018):
Balancing cellular redox metabolism in microbial electrosynthesis and electro fermentation – A chance for metabolic engineering
Metab. Eng. 45 , 109 - 120
10.1016/j.ymben.2017.12.003 - Yu, S., Lai, B., Plan, M.R., Hodson, M.P., Lestari, E.A., Song, H., Krömer, J.O. (2018):
Improved performance of Pseudomonas putida in a bioelectrochemical system through overexpression of periplasmic glucose dehydrogenase
Biotechnol. Bioeng. 115 (1), 145 - 155
10.1002/bit.26433