Hazard Assessment of Ionizable Organic Chemicals
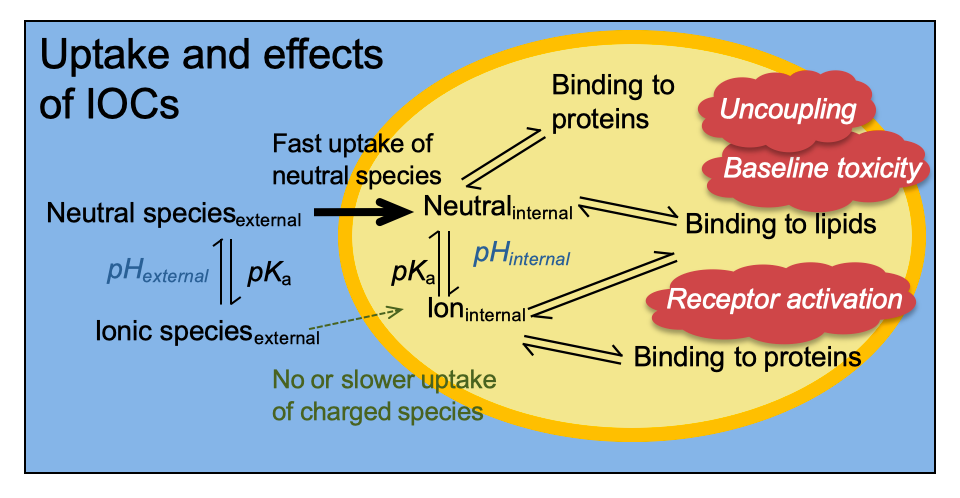
Many chemicals in commerce are ionizable organic chemicals (IOCs), including pesticides, pharmaceuticals and ingredients of personal care products and food stuff. IOCs change they speciation depending on the pH value of the surrounding environment and can either have one (monoprotic acids and bases) or multiple ionizable functional groups (multifunctional and zwitterionic IOCs).
The speciation of IOCs is most important for their uptake into organisms as the charged species is taken up slower and to a lesser degree compared to the neutral species. Inside the organism, where the pH is buffered, the internal effect concentration is constant. Our group is using ion-trapping models to predict the internal concentrations of IOCs.
We are also interested in the pH-dependence of toxicity of IOCs. The most important speciation-dependent mode of action is uncoupling, where both – neutral and charged – species have to act together to transport protons across the energy-transducing membrane, destroying the ion gradient.
While the majority of IOCs turned out to be baseline toxicants if the speciation is properly considered, the development of ion trapping models and baseline toxicity QSARs for IOC helps to unambiguously classify the effects of IOCs.
