Archive Department News
June 2025
Team News
What an exciting week at SETAC Europe 2025 in Vienna!
We are very pleased that we were able to present our current research work both in presentations and at the Poster Corner. It was
great opportunity to share our results with the international community community and to receive valuable feedback.
Particularly inspiring were the numerous sessions on New Approach Methodologies (NAMs) and the assessment of the chemical exposome - topics that are topics that are more topical than ever and provide important impetus for future research.
In addition to all the exciting scientific content, it was also a wonderful opportunity to meet up with old acquaintances, make new contacts and exchange ideas in a exchange ideas in a personal setting.
Present were: Elena Hommel, Naroa Lopez, Vanessa Srebny & Julia Huchthausen - a great team with a passion for environmental research and innovative methods.
Elena Hommel, Naroa Lopez, Vanessa Srebny & Julia Huchthausen
February 2025
Team News
Welcome to our new PhD student Yuxin Liu in the QTOX project. Yuxin started with the QTOX annual meeting in Valencia where she (second from left) and other QTOX doctoral candidates inspected the mesocosms for our joint QTOX mesocosm study in 2026. Yuxin’s project will deal with quantitative in vitro to vivo extrapolations from cells to fish.



November 2024
Team News
The Department of Cell Toxicology spent two days at the beginning of November on a departmental retreat at the beautiful Lake Störmthal. In addition to detailed presentations on the various current projects and lively discussions on the scientific future of the department, there were also fun opportunities for the team to grow together. 
June 2024
New partnership
We will start a collaboration with Prof., Kathrin Fenner at Eawag and Uni Zürich and Prof. Kjell Jorner of ETHZ with a a new project funded by the Swiss National Science Foundation on the development of environmentally friendly chemicals at the crossroads of chemical, computer, environmental and health science. More info at LinkMay 2024
Staff News
Warmest gratulations to Dr. Weiping Qin for successfully defending her PhD thesis on exposure and toxicity of PFAS at the University of Tübingen.
Two in a day: Zelltox congratulates the two “external” members of the team Dr. Isis Roemer and Dr. Ran Wei for successfully defending their doctoral theses at the University of Tübingen.

April 2024
Team News
We welcome Haotian Wang from Beijing for a two-year postdoc sponsored by the Humboldt foundation. Haotian gave a poster presentation about his project at the Humboldt Network Meeting in Magdeburg from 24 to 26 April. In his project at UFZ, he will identify neurotoxic chemicals in the aquatic environment by integrating data science with impact-oriented analysis using high-throughput bioanalytical tools.

Hot Papers
Congratulations to Weiping Qin for designing an awesome front cover for this weeks ES&T (link) together with an excellent article on the baseline toxicity prediction of PFAS toxicity and identification of specific effects (link)

Staff News
We farewell Dr. Jungen Lee who worked with Celltox for 6 years, completing a PhD and a postdoc and bringing us up to speed with imaging in high-throughput assays. On the day of her farewell a new paper appeared in ES&T on a novel high throughput assay that combines mitochondrial toxicity and oxidative stress response (link) with applications for risk assessment of chemicals and water quality monitoring. All the best to you Jungeun for your future endeavours. We will miss you!

March 2024
Staff News
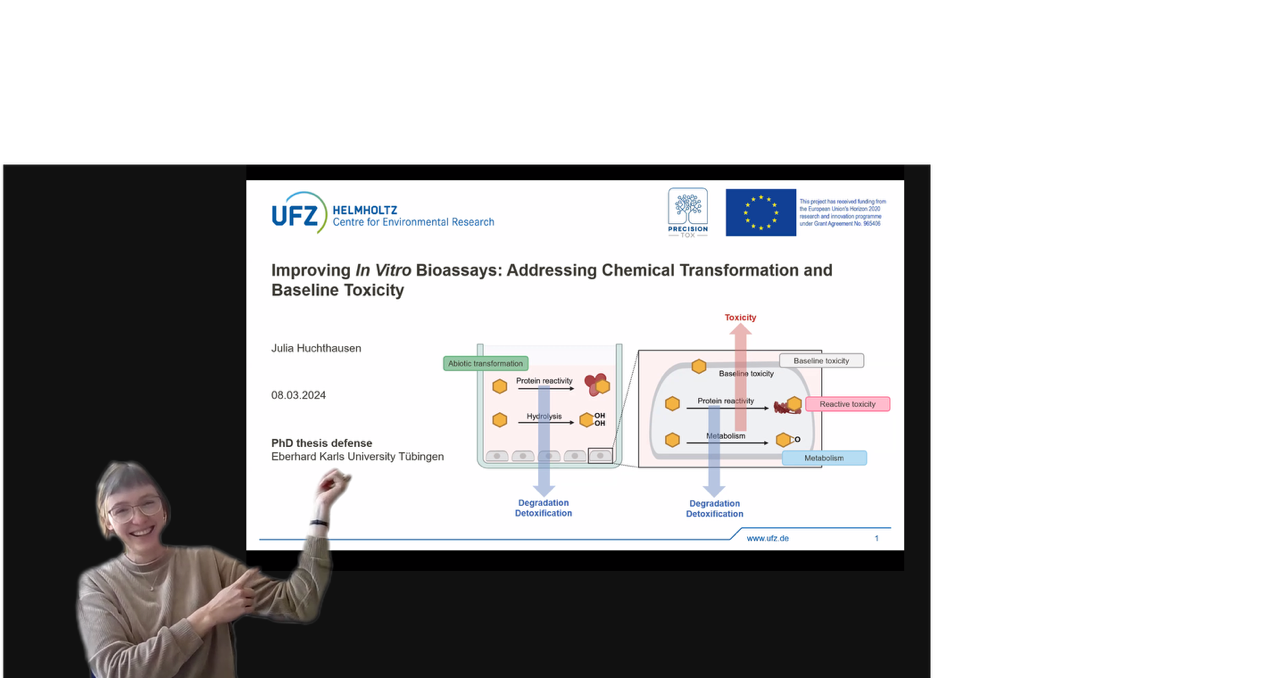
Congratulations to our Julia Huchthausen on successfully passing her doctoral thesis defence.
Your hard work, perseverance and passion for research have paid off. Not only have you achieved an intellectual feat, but you have also taken an important step in your scientific career.


January 2024
New Partnership
We will start a collaboration with Prof., Kathrin Fenner at Eawag and Uni Zürich and Prof. Kjell Jorner of ETHZ with a a new project funded by the Swiss National Science Foundation on the development of environmentally friendly chemicals at the crossroads of chemical, computer, environmental and health science.
February 2024
Hot Paper
First paper published stemming from a collaboration project between the University of Queensland and UFZ funded by DAAD
Bade R, Huchthausen J, Huber C, Dewapriya P, Tscharke BJ, Verhagen R, Puljevic C, Escher BI, O'Brien JW. 2024. Improving wastewater-based epidemiology for new psychoactive substance surveillance by combining a high-throughput in vitro metabolism assay and LC−HRMS metabolite identification.
Water Res 253:121297.
November 2023
Hot Paper
Georg Braun published the extraction method for human plasma preparing future human biomonitoring studies using target analysis of 400 chemicals and in vitro bioassays in ES&T- link
Conference
Zelltox PhD student Georg Braun and department leader Beate Escher traveled to the ISEAC-41 in Amsterdam and presented on their work on in vitro bioassays in mixture effect assessment, extraction methods of difficult matrices and human biomonitoring.


July 2023
Conference
Zelltox PhD student Weiping Qin presented her first paper on the role of protein binding of PFAS in cell-based bioassay at the SETAC Europe 2023 conference in Dublin and at the JRC summer school on new approach methods in Ispra.
 Weipings talk at SETAC
Weipings talk at SETAC
Hot Paper
We are always pleased to host young scholars at Celltox for measurement of their samples in in vitro assays. Check out two new papers stemming from such collaborations.
Dr. Pavel Saur is a fish biology expert who worked on quarter quality during his visit at UFZ and his paper "Bioanalytical and chemical characterization of organic micropollutant mixtures in long-term exposed passive samplers from the Joint Danube Survey 4: Setting a baseline for water quality monitoring” is a first testimony of this fruitful collaboration.
Fei Chen visited from Guangdong Key Laboratory of Environmental Pollution and Health, School of Environment, Jinan University, Guangzhou, China (before the pandemic) and first outcomes can be found in "Text Mining-Based Suspect Screening for Aquatic Risk Assessment in the Big Data Era: Event-Driven Taxonomy Links Chemical Exposures and Hazards”
June 2023
Hot Paper
Fancy travel to Barcelona? All you need to know about drinking water quality in all suburbs of Barcelona….
Your article is available on nature.com
March 2023
Publication
Together with a large consortium of researchers from Helmholtz and international universities we have published a concept for modernising the hazard assessment of chemicals. The proposal is to combine effect and degradation assessment in a common approach using in vitro bioassays and new approach methods
Publication
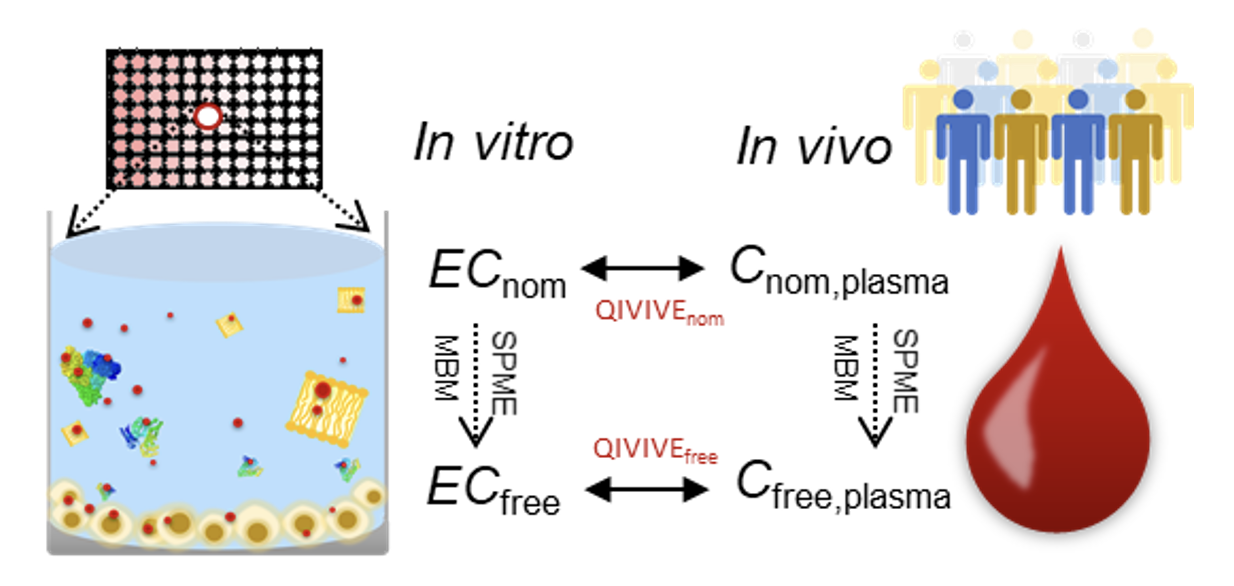
Congratulations to Weiping Qin for publishing her first PhD paper. If you want to know why PFAS behave differently in in vitro bioassays and in human blood, check out " Role of bioavailability and protein binding of four anionic perfluoroalkyl substances in cell-based bioassays for quantitative in vitro to in vivo extrapolations“
Award
Prof. Bryan Brooks , Editor-in Chief of Environmental Science and Technology letters presented the “Outstanding Achievements in Environmental Science and Technology Awards” to our dpartment Head Prof. Beate Escher and to Prof. Urs von Gunten of Eawag/EPFL, Switzerland. Beate talked in her award lecture about "Learning from environmental chemistry: a mechanism-based approach to toxicology and chemical risk assessment”


February 2023
virtuell Publication
Our very own Luise Henneberger (#LuiHenneberger) is a contributor to the virtual issue celebrating the international day of women and girls in science #ChemResTox

January 2023
Hot Paper
We congratulate Georg to his first PhD paper entitled Prioritization of mixtures of neurotoxic chemicals for biomonitoring using high-throughput toxicokinetics and mixture toxicity modeling. Stay tuned for new discoveries in biomonitoring thanks to this modelling study.
Hot Paper
Our guest from ETH, Juliane Glüge published the product our our collaboration as guest scientist at UFZ, an analysis of the bioaccumulation data of the ECHA databases entitled "How error-prone bioaccumulation experiments affect the risk assessment of hydrophobic chemicals and what could be improved”
December 2022
Conference
Jungeun Lee and Julia Huchthausen joined the 21st International Congress ESTIV in Sitges (Barcelona) at the end of November which is focused on the development of in vitro and in silico approaches.
Jungeun Lee presented her poster about mixture effects of extracts from environment, food and blood on neurite outgrowth compared to cytotoxicity in SH-SY5Y cells.
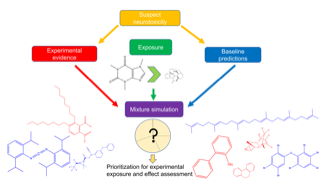
Julia Huchthausen got the opportunity to give a presentation about a high-throughput workflow for the assessment of the abiotic stability of chemicals in in vitro bioassays.

November 2022
Recruiting
Join our team at UFZ Cell Toxicology for this exciting opportunity (PhD Researcher/Topic: New Hazard Indicators for the Evaluation of Replacement Products (m/f/x) ) to modernize PBT-assessment within environmental risk assessment of chemicals. A Master in environmental sciences, biology, chemistry, bioanalysis, (eco)toxicology, environmental system analysis, geoecology or related areas is wanted!
@UFZ_CITE
October 2022
Publication
While our first paper (link to https://www.mdpi.com/1660-4601/19/20/12990) of the H2020 Project Panoramix is out - Mixture Risk Assessment of Complex Real-Life Mixtures—the PANORAMIX Project has just appeared, the project is already in full swing with the first General Assembly having taking place from 6-7 October 2022 in the Comics Museum in Brussels
Team News
We farewell Rita Schlichting, who helped build up CITEPro for over five years and did magic with lab management, and bioassay development including automatic data evaluation. We will miss you and wish you all the best for your new position outside academia.

September 2022
After three years and uncountable Teams meetings (with only the kick-off and now the final workshop in person) we had the grand finale of the Global Water Research Coalition project “Effect-based monitoring in water safety planning on 19 September 2022 in Paris. What a happy and industrious team- KWR, Griffith Uni, UFZ as well as Suez and Veolia as industry partners. Numerous reports can be downloaded on http://www.globalwaterresearchcoalition.net/ and several papers forthcoming. Hopefully more to come.

July 2022
Wholehearted congratulations to Dr. Jungeun Lee for successfully defending her PhD thesis entitled" Development of high-throughput methods for testing neurotoxicity of environmental samples” Awesome outcome!

May 2022
A delegation of Celltoxies and external collaborators presented their work on cell-based bioassays at SETAC 2022 in Copenhagen - a few impressions:

April 2022
Much of our work is on the speciation and toxicity of ionisable organic chemicals but for once we teamed up with environmental fate experts and contributed to the ES&T Feature Article "Sorption and Mobility of Charged Organic Compounds: How to Confront and Overcome Limitations in Their Assessment.“ Thanks to Gabriel Sigmund for leading this effort that brought together a large group of authors: Gabriel Sigmund,Hans Peter H. Arp, Benedikt M. Aumeier, Thomas D. Bucheli, Benny Chefetz,
Wei Chen, Steven T. J. Droge, Satoshi Endo, Beate I. Escher, Sarah E. Hale, Thilo Hofmann,
Joseph Pignatello, Thorsten Reemtsma, Torsten C. Schmidt, Carina D. Schönsee, and Martin Scheringer.
February 2022
Publication
Jungeun Lee and coworkers established a robust neuronal toxicity assay that allows not only classification of neurotoxicants for specificity of neurite outgrowth inhibition, neurotoxicity and baseline toxicity but will also be applied on the future in environmental monitoring and biomonitoring.
Project
Winner! We are part of the winning team of Phase 2 of the CRACK IT Challenges 36!
The project CRACK IT Challenge 36: Animal-free in vitro led by Victoria Hutter of ImmuOne aims to develop animal product-free adaptations of the in vitro OECD test guidelines TG487 and TG455 to improve human-relevant responses and reproducibility.
Our role in the project is the characterization and validation of the metabolic activity of the S9 alternative and the participation in the interlaboratory validation.
December 2021
The Project PANORAMIX (Providing risk assessments of complex real-life mixtures for the protection of European citizens and the environment) was kicked off with a meeting at the Technical University of Denmark, who is coordinating this EU Horizon 2020 project. The Department Cell Toxicology leads the work package on in vitro bioassays for biomonitoring.

September 2021
Our congratulations go to Nadine Sossalla from UBZ, who successfully defended her PhD thesis on 23 September 2021. Nadine – it was a pleasure working with you despite the hundreds of samples you swamped our lab with. You are our truly first high-throughput student. All the best for your postdoc in Norway.

June 2021

10 years in the thinking, 1 year in the writing – check out the open-access second edition of “Bioanalytical Tools in Water Quality Assessment” and supplementary material.
Thank you to our coauthors Dr. Peta Neale, long-time partner in crime, and Prof. Fred Leusch, who introduced us to working with human cell lines.
May 2021
If you cannot travel in person, travel virtually: Beate presented at the Northwest Fischeries Science Center in Seattle about application of bioassays for water quality monitoring.

Congratulations to Dr. Fabian Fischer, who was awarded the “Promotionspreis auf dem Gebiet der Wasserchemie” 2020 for his PhD dissertation, which he presented at the virtual “Wasser 21” conference.
April 2021
We welcome three new team members in March 2021. Weiping Qin joined us with a Chinese Scholarship Council grant to do her PhD on the topic of in vitro toxicology and exposure assessment of perfluorinated compounds. Dr. Andrea Gärtner joined as a postdoc to work on mixtures on metals and organics and Fritz Kramer will do research towards his Master thesis in Molecular Medicine at the University of Jena.
March 2021
Our Master Student Audrey Ogefere from Potsdam University, who was supervised by Luise Henneberger, defended her Master thesis "The pH-Dependent Toxicity of Pharmaceuticals in In Vitro Cell-based Bioassays” successfully in a Zoom meeting on 5 March 2021- although we could not be with you, we send our congratulations via the web!

November 2020
We congratulate Max Müller to successfully passing his doctoral exam at University of Tübingen with his thesis “Organic micropollutants in small river systems – Occurrence, fate and effect”. He performed the effect part of his study in the department Cell Toxicology at UFZ.

September 2020
We had our department retreat in a bit of simplified way this year to comply with the COVID-19 hygiene measures. We explored each department member’s career trajectory and had some fun with mini-golf in the afternoon.


August 2020
Due to COVID-19, the CAMPOS International Conference 2020 "Turnover of Diffuse Pollutants on the Catchment Scale” had to be cancelled but a virtuel poster session of the CAMPOS Conference was launched where you can also find contributions from Celltox in collaboration with Tübingen University.
July 2020
Since July, 1st, Julia Huchthausen, Luise Henneberger and Beate Escher are working in a third-party funded project financed by Unilever on the development of in vitro testing strategies for unstable chemicals.
Mai 2020
The Global Water Research Coalition (GWRC) project on Effect Based Monitoring (EBM) in Water Safety Planning was started in October 2019 and UFZ is responsible for the Workpackage 3, on applicability of EBM tools and methods. The first deliverable from the project, a factsheet on “the use of effect-based monitoring for the assessment of risks of low-level mixtures of chemical in water on man and the environment” is now available on the GWRC website.
March 2020
Due to the pandemic our team has also moved to home office to make our contribution to stop the spreading of COVID19. We can be reached by e-mail and via videoconferencing. While chemical pollution is not in the centre of attention today, it remains an ongoing issue and our work will continue to contribute to a future without chemical pollution.
February 2020
Read the new Science special issue on "Chemistry for tomorrow’s earth” on sciencemag.org with a contribution from our department on “Tracking complex mixtures of chemicals in our changing environment" Beate I. Escher, Heather M. Stapleton, and Emma L. Schymanski (2020): Tracking Complex Mixtures of Chemicals in our Changing Environment. Science, https://doi.org/10.1126/science.aay6636
and listen to the podcast on https://www.sciencemag.org/podcast/getting-bisphenol-out-food-containers-and-tracing-minute-chemical-mixtures-environment
January 2020
We farewell Dr. Annika Jahnke and her team who will move on to form an independent group in the Department of Ecological Chemistry and thank her and her team for the fruitful collaboration over the last five years. At the same time we welcome Dr. Luise Henneberger as the new group leader of the team “Bioassay exposure assessment”.
September 2019
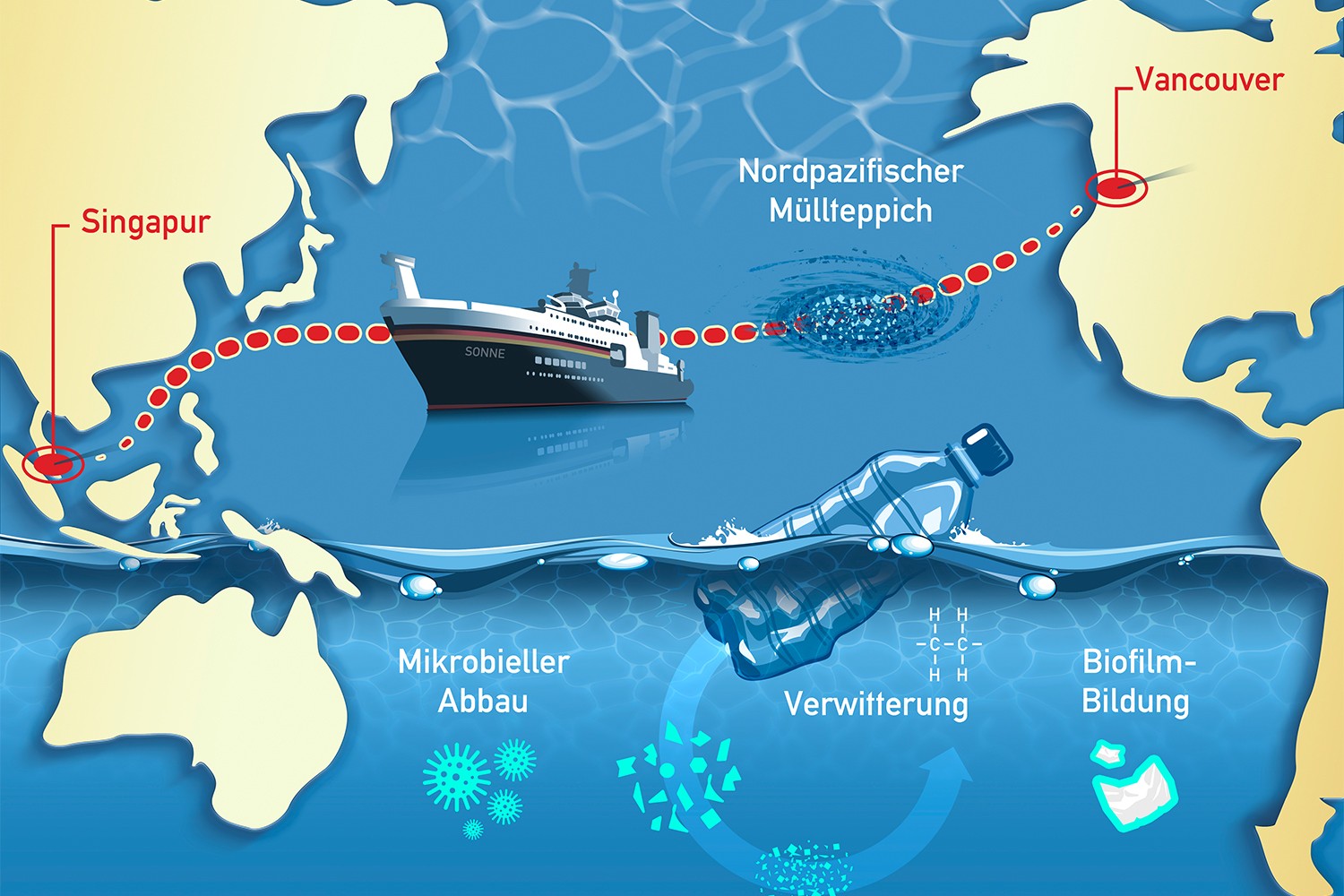
Annika, Elisa, Christoph and Cedric arrived safely back in Singapore after their expedition across the Pacific. Read more about it in the blog. More information on the project Micro_fate can be found here.
August 2019
We farewell Jenny into her maternity leave and wish her all the best for the future!

July 2019
Luise Henneberger has published a method for experimental determination of partitioning of organic acids in biological materials: “C18-coated solid-phase microextraction fibers for the quantification of partitioning of organic acids to proteins, lipids, and cells”
https://pubs.acs.org/doi/10.1021/acs.chemrestox.8b00249
June 2019
Our department is growing:
Sebastian Abel joined the team as a postdoc in April. He studies the human exposure using passive sampling devices.
Lili Niu is a scholarship holder of the Alexander-von-Humboldt Foundation and works on the bioavalability and toxicity of sediment-associated pollutants.
Welcome to the team!
May 2019
Department Head Beate Escher visited several Chinese Universities in April, among them Jinan University, Shanghai Ocean University and Being Normal University to deepen existing collaborations and for scientific
exchange.

April 2019
Department Head Beate Escher giving lectures on environmental and human heath risk assessment at Being Normal University during her trip to China.