CHEMO-RISK -
Chemometers for in situ risk assessment of mixtures of pollutants

The ERC-funded project CHEMO-RISK started in May, 2017. CHEMO-RISK aims for a novel scientifically sound chemical risk assessment paradigm that integrates exposure and effect assessment of a broad range of chemicals into a single procedure and provides information relevant to ecosystem and human health. The key innovation is polymer “chemometers” that will be equilibrated with their surroundings and deliver information on the pollutant’s chemical activity in the environment, biota, and humans. A chemometer functions analogously to a thermometer, but instead of the temperature, it yields a measure of chemical activity. Chemical activity in turn indicates the thermodynamic potential for, e.g., partitioning, biouptake and toxicity. CHEMO-RISK aims at breaking the current paradigm in environmental risk assessment of single chemicals that disregards bioavailability, ignores mixture effects, lacks site-specificity and is difficult to extrapolate to human health
Group leader: Annika Jahnke (contact: annika.jahnke[at]ufz.de)
Group co-lead: Elisa Rojo Nieto (contact: elisa.rojo-nieto[at]ufz.de)
Former team members: Eva Reiter, Jörg Watzke, Theo Wernicke, Meliz Muz, Sebastian Abel, Fanny Rosello, Veronika Schacht, Njoud Alsabbagh, Hanna Nolte, Madlen Landmann, Anne Jäger, Anna Sophia Martin
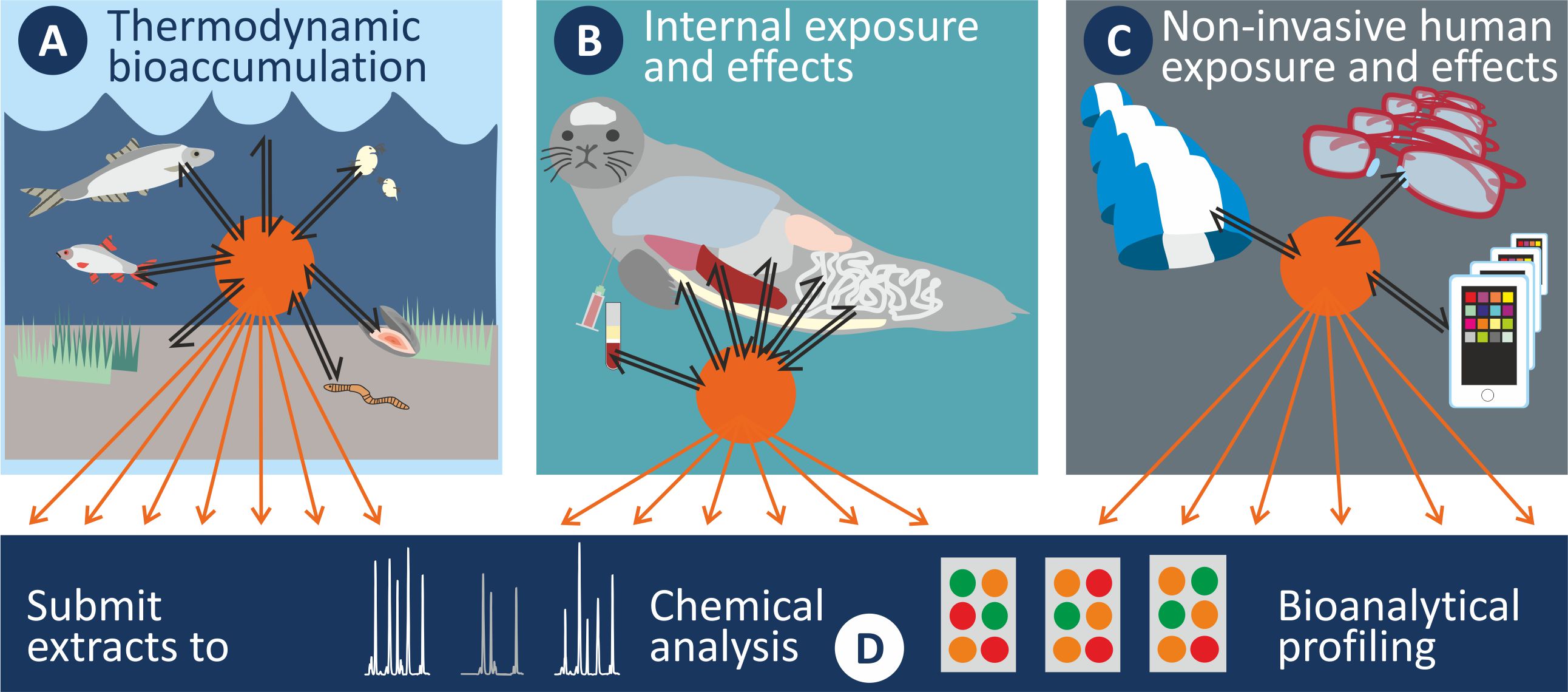
CHEMO-RISK aims for a novel scientifically sound chemical risk assessment paradigm that integrates exposure and effect assessment of a broad range of chemicals into a single procedure and provides information relevant to ecosystem and human health. The overall objectives are to answer the following questions: Which processes drive the enrichment of pollutants in aquatic biota on a thermodynamic basis (subproject A, Elisa Rojo Nieto)? How do pollutants distribute within an organism, and which effects do they elicit at the key target sites (subproject B, Eva Reiter)? Can we apply everyday-life items such as silicone plasters to replace invasive sampling of blood in human health risk assessment (subproject C, Sebastian Abel)? To which degree can pattern analysis of chemometer extracts help identify emerging chemicals and explain the observed toxicity profiles across media (subproject D, Veronika Schacht)?
Publications (available from https://zenodo.org)
[1] Rojo-Nieto, E.; Muz, M.; Koschorreck, J.; Rüdel, H.; Jahnke, A. Passive equilibrium sampling of hydrophobic organic compounds in homogenised fish tissues of low lipid content. Chemosphere 2019, 220, 501. https://doi.org/10.1016/j.chemosphere.2018.12.134
[2] Reiter, E.B.; Jahnke, A.; König, M.; Siebert, U.; Escher, B.I. Influence of Co-Dosed Lipids from Biota Extracts on the Availability of Chemicals in In Vitro Cell-Based Bioassays. Environ. Sci. Technol. 2020, 54, 4240. https://doi.org/10.1021/acs.est.9b07850
[3] Muz, M.; Escher, B.I.; Jahnke, A. Bioavailable Environmental Pollutant Patterns in Sediments from Passive Equilibrium Sampling. Environ. Sci. Technol. 2020, 54, 15861. https://doi.org/10.1021/acs.est.0c05537
[4] Muz, M.; Rojo-Nieto, E.; Jahnke, A. Removing Disturbing Matrix Constituents from Biota Extracts from Total Extraction and Silicone-Based Passive Sampling. Environ. Toxicol. Chem. 2021, 40, 2693. https://doi.org/10.1002/etc.5153
[5] Wernicke, T.; Abel, S.; Escher, B.I.; Koschorreck, J.; Rüdel, H.; Jahnke, A. Equilibrium sampling of suspended particulate matter as a universal proxy for fish and mussel monitoring. Ecotoxicol. Environ. Safe. 2022, 232, 113285. https://doi.org/10.1016/j.ecoenv.2022.113285
[6] Reiter, E.B.; Escher, B.I.; Siebert, U.; Jahnke, A. Activation of the xenobiotic metabolism and oxidative stress response by mixtures of organic pollutants extracted with in-tissue passive sampling from liver, kidney, brain and blubber of marine mammals. Environ. Int. 2022, 165, 107337. https://doi.org/10.1016/j.envint.2022.107337
[7] Wernicke, T.; Rojo-Nieto, E.; Paschke, A.; Nogueira Tavares, C.; Brauns, M.; Jahnke, A. Exploring the partitioning of hydrophobic organic compounds between water, suspended particulate matter and diverse fish species in a German river ecosystem. Environ. Sci. Europe 2022, 34, 66. https://doi.org/10.1186/s12302-022-00644-w
[8] Rohner, S.; Morell, M.; Wohlsein, P.; Stürznickel, J.; Reiter, E.B.; Jahnke, A.; Prenger-Berninghoff, E.; Ewers, C.; Walther, G.; Striewe, L.C.; Failla, A.V.; Siebert, U. Fatal aspergillosis and evidence of unrelated hearing loss in a harbor porpoise (Phocoena phocoena) from the German Baltic Sea. Front. Mar. Sci. 2022, https://doi.org/10.3389/fmars.2022.958019
[9] Rojo-Nieto, E.; Jahnke, A. Chemometers: an integrative tool for chemical assessment in multimedia environments. Chem. Comm. 2023, 59, 3193. https://doi.org/10.1039/D2CC06882F
[10] Reiter, E.B.; Escher, B.I.; Rojo-Nieto, E.; Nolte, H.; Siebert, U.; Jahnke, A. Characterizing the marine mammal exposome by iceberg modeling, linking chemical analysis and in vitro bioassays. Environ. Sci.: Processes Impacts 2023, Advance Article, https://doi.org/10.1039/d3em00033h

The CHEMO-RISK team has addressed developing ”chemometer” passive samplers for the transfer of mixtures of environmental pollutants from different environmental media (sediment, water, biota of various trophic levels) and body compartments (blubber, brain, liver, kidney and blood of mammals) to the laboratory for their characterization. A special focus of the ”chemometer” concept is on comparability across media which is achieved by using silicone as the common sampling phase for all types of samples. The pollutants partition into the silicone according to their activity in the sample until both phases reach thermodynamic equilibrium. Then, the silicone is retrieved, its surface is thoroughly cleaned and the chemicals are solvent-extracted for chemical analysis or bioanalytical screening. The data (equilibrium partitioning concentrations in silicone, Csilicone⇌medium) can then be compared directly across media, e.g., to assess biota/sediment accumulation of various pollutants.
The issue of mixture risk characterization is of huge importance for society which on the one hand largely relies on multiple chemicals to provide or secure certain service functions (e.g., food, clean water, transport, medication), while on the other hand striving for a healthy, non-toxic environment for humans and ecosystems. Fulfilling both requirements calls for sound assessment of the hazards and risks arising from the presence of complex mixtures of chemicals in the multimedia environment covering a multitude of different ecosystems and environmental conditions. However, current risk assessment relies on a small number of individual, so-called ”indicator” chemicals being monitored and their effect testing in few indicator species, which implies uncertainty in terms of potential cocktail effects of multiple chemicals and does not cover diverse ecosystems where these chemicals may, e.g., be bioavailable to different degrees.

