Material developments for water treatment
The development of novel materials for water treatment is one of our main research fields. This includes materials for adsorption or degradation of organic contaminants (catalysts and reagents) as well as the combination of both processes. Especially for the treatment of micropollutants in water degradation after adsorptive enrichment is a promising approach.
In drinking water or wastewater treatment pelletized materials in fixed beds are frequently used. At the same time, micro- or nanoscale particles are of special interest for in-situ groundwater treatment. These fine particles can be injected in the form of suspensions into contaminated aquifers where they are deposited on sediment, forming a reactive zone which removes contaminants from passing groundwater (nanoremediation). Particles for nanoremediation must be optimized not only regarding efficiency for contaminant removal but also their suspension and transport properties. In addition, they need to fulfil strict requirements regarding environmental impact. Our material developments are:
Carbo-Iron®
Carbo-Iron, a composite material consisting of colloidal activated carbon and 20-30 wt% embedded nanoiron structures, was initially developed to overcome limitations of pure iron nanoparticles. Two environmentally friendly materials (C and Fe) were combined in an optimized way to generate beneficial properties, such as improved subsurface mobility, enrichment of pollutants directly in the reactive grain (trap & treat principle), long-term stability of the iron by suppressed corrosion and support of subsequent biological clean-up activities in the aquifer. With these properties, Carbo-Iron became an environemnetally friendly in-situ reagent which is suitabel for plume and source treatment.
Trap-Ox® Fe-Zeolites
Trap-Ox® Fe-zeolites is a particle family of Fe-zeolites tailored for in situ trapping of organic contaminants by adsorption and catalytic oxidation in combination with oxidants such a hydrogen peroxide. These microporous mineral adsorbent particles form stable suspension which can be injected into the aquifer. After deposition on aquifer sediment they act as adsorbents which immediately stop any further spreading of contaminants. This sorption zone can be repeatedly regenerated by injection of H2O2 solution whereby iron species in the zeolite act as Fenton-like catalysts.
Colloidal Activated Carbon
Oxidation catalysts for wastewater treatment
Catalysts for reductive dehalogenation
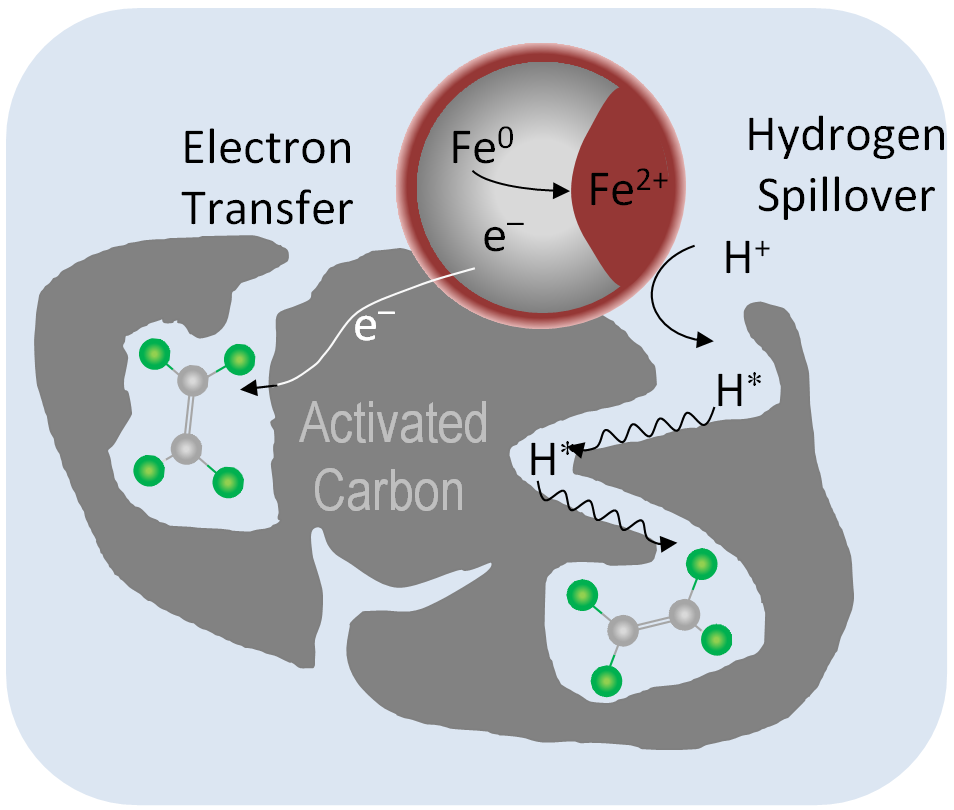
Palladium is an efficient but at the same time very sensitive catalysts which enables dehalogenation of a wide variety of water pollutant under mild conditions using hydrogen or hydrogen donors. As alternative to the poison sensitive palladium, the much more stable copper is able to use borohydride as reducing agent and catalyze dehalogenation of alophatic and aromatic pollutants. We search for suitable methods to enable transition metal catalysts for their utilization under environmental conditions and optimize life-time and selectivities of dehalogenation catalysts.

