Press Release, 23. April 2026
Efficient degradation of short-chain PFAS
UFZ team develops new method to remove perfluorobutanoic acid from water
Short-chain perfluorinated and polyfluorinated alkyl compounds (PFAS) such as perfluorobutanoic acid (PFBA) are increasingly entering the environment via various pathways and contaminating groundwater and drinking water. Because PFAS are highly mobile, removing them has so far required a great deal of effort. But a research team at the Helmholtz Centre for Environmental Research (UFZ) has developed a new technology to do so. According to an article recently published in Chemical Engineering Journal, the new process is more environmentally friendly and less energy-intensive.

Short-chain PFAS, such as perfluorobutanoic acid, enter the environment through firefighting foams, for example, and degrade only very slowly if at all.
Photo: burnstuff2003_AdobeStock
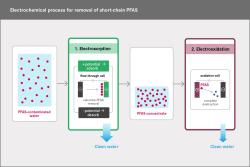
During this process, the PFAS are first separated and concentrated by electrosorption (Step 1) and then destroyed by electrooxidation (Step 2). The main byproducts are CO2 and fluoride.
Photo: Susan Walter-Pantzer / UFZ (graphic editing)
About 10,000 PFAS substances are currently known, of which 4,000–5,000 are used in industry, including in the production of outdoor clothing, food packaging, cookware, and cosmetics. Numerous PFAS – such as those found in firefighting foams – enter the environment and degrade only very slowly if at all. Because they pose a risk to human health by affecting metabolism, hormone balance, reproduction, and the immune system and are suspected of being carcinogenic, many long-chain PFAS have been regulated under the Stockholm Convention. Their production and use are now prohibited or restricted. In response, they have been increasingly replaced with short-chain PFAS. As a result, compounds such as perfluorobutanoic acid (PFBA) are being detected more frequently in the environment. With only four carbon atoms and a carboxyl group at one end of the molecule, PFBA strongly attracts water. "That is why PFBA dissolves readily in water and is highly mobile. It is therefore difficult to remove PFBA from water using conventional methods such as activated carbon adsorption", says Dr Anett Georgi, UFZ chemist and co-author.
To remove PFBA from water, the UFZ research team has developed a two-stage electrochemical purification process in which PFBA is first concentrated and then broken down. How it works: In the first step, large volumes of PFBA-containing water are passed through a flow cell with an electrode made of a textile-like activated carbon fibre felt; this electrode is given a slight positive charge for electro-adsorption. "This causes the negatively charged PFBA to accumulate on the surface of the activated carbon", says Dr Navid Saeidi, UFZ environmental engineer and lead author. By reversing the polarity of the voltage, the PFBA is then detached from the surface, rinsed away with a small volume of water, and collected as a concentrate. This can increase the PFBA concentration by a factor of 40. By arranging the electro-sorption cells in a cascade configuration, this enrichment process can be repeated several times. In the second step, PFBA is broken down by electro-oxidation at a boron-doped diamond electrode (i.e. through a chemical purification of the water triggered by an electric current). The anode has a strong oxidising effect and causes PFBA to decompose. The main by-product is fluoride, which is easily separated.
"All steps can be carried out on site, thereby reducing transport costs and energy requirements", says Georgi. Because PFBA adsorption is controlled by applying an electrical voltage, the activated carbon material can be regenerated repeatedly and reused multiple times – unlike other processes in which PFAS-contaminated activated carbon must be disposed of in waste incineration plants or regenerated with high energy input. "This not only conserves fossil resources but also reduces CO2 emissions because activated carbon is often produced from hard coal and is mainly imported from Asia", says Georgi.
The UFZ scientists have already filed a patent for this process because they see many potential applications, particularly in removing PFAS from municipal and industrial wastewater streams, including at airports, where groundwater is contaminated with short- and long-chain PFAS as a result of the use of firefighting foam. "In light of the increasingly stringent PFAS limits that operators are required to comply with, there is a need for more efficient removal technologies that are as reliable, environmentally friendly, and affordable as our method. It could complement traditional activated carbon adsorbers in cases of complex PFAS contamination and capture short-chain PFAS", says Dr Katrin Mackenzie, UFZ chemist and co-author. This would result in a considerably longer service life for the entire adsorber unit and thus lead to cost savings.
Publication:
Navid Saeidi, Sarah Sühnholz, Katrin Mackenzie, Anett Georgi: A two-step electrochemical approach for an efficient destruction of short-chain PFAS in water, Chemical Engineering Journal, https://doi.org/10.1016/j.cej.2026.172856
Further information
Dr Anett Georgi
UFZ Department of Technical Biogeochemistry
anett.georgi@ufz.de
Dr Katrin Mackenzie
Head of the UFZ Department of Technical Biogeochemistry
katrin.mackenzie@ufz.de
UFZ press office
Susanne Hufe
Phone: +49 341 6025-1630
presse@ufz.de
In the Helmholtz Centre for Environmental Research (UFZ), scientists conduct research into the causes and consequences of far-reaching environmental changes. Their areas of study cover water resources, ecosystems of the future, environmental technologies and biotechnologies, the effects of chemicals in the environment, modelling and social-scientific issues. The UFZ employs more than 1,100 staff at its sites in Leipzig, Halle and Magdeburg. It is funded by the Federal Government, Saxony and Saxony-Anhalt.
www.ufz.deThe Helmholtz Association contributes to solving major challenges facing society, science and the economy with top scientific achievements in six research fields: Energy; Earth and Environment; Health; Key Technologies; Matter; and Aeronautics, Space and Transport. With some 39,000 employees in 19 research centres, the Helmholtz Association is Germany’s largest scientific organisation.
www.helmholtz.de